Research
Cardiovascular-Immunology
The CSL Cardiovascular-Immunology research program is working to understand the crosstalk between the cardiovascular and the innate and adaptive immune system in the scope of acute myocardial infarction, heart failure, myocarditis and atherosclerosis with the goal of developing therapeutic options.
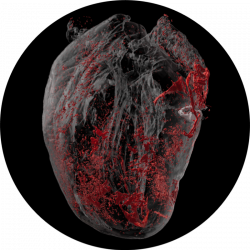
Our immune system plays an important role in the cardiovascular system under both homeostatic and perturbed conditions. In response to infection or after myocardial infarction, large numbers of immune cells are recruited to the heart to scavenge pathogens, remove dying tissue and promote healing. Under some circumstances, immune cells can cause irreversible damage, contributing to heart failure. Also, various innate and adaptive immune pathways have been identified in atherosclerosis.
Cardioprotection – Mitochondrial Physiopathology
The CardioScienceLabs Mitochondrial Physiopathological research program is working to understand the molecular pathways that regulate mitochondrial decision on cell fate with the goal of developing a protection shield to ensure mitochondrial health, preventing loss of cardiac function.

Mitochondria are multifunctional organelles, and their structural and functional integrity is fundamental to cell life. They can drive cellular dysfunction and demise. Mitochondria supply ATP through oxidative phosphorylation and a major hub for cellular Ca2+ signaling. They can be restricted in ATP synthesis and can become the major source of reactive oxygen species. Mitochondria represent a point of convergence for a variety of upstream cell death stimuli and subsequent transmission of signals to downstream executioner proteins.
Cardio-Oncology
The CSL Onco-Cardiology research program is working to understand the roots of cardiotoxicity with the goal of developing cardioprotective therapies for a rapidly growing population of cancer survivors.

One of the most important consequences of chemotherapy are adverse effects on the heart’s function, leading to an increase in morbidity and mortality of cancer survivors.
Microbiome
The CardioScienceLabs Microbiome research program seeks to gain a deeper understanding of the underlying mechanisms to find new ways of microbiome modulation to salvage the burden of acute cardiovascular events.
A growing body of data shows a mutual interaction between the oral/enteral microbiome and acute cardiovascular diseases. Depending on external factors, microbiome derived metabolites can aggravate or reduce acute cardiovascular pathologies.
Arrhythmias & Cardiac Innervation
The CSL Arrhythmia & Cardiac Innervation research program is working to define the influence of the autonomic nervous system and cardiac inflammation on ventricular arrhythmogenesis.

We are engaged in research on the relevance of sympathetic innervation on ventricular arrhythmogenesis, with the goal of improving therapeutical options for patients with ischemic and non-ischemic cardiomyopathy.
Research partners around the globe
Our Partners